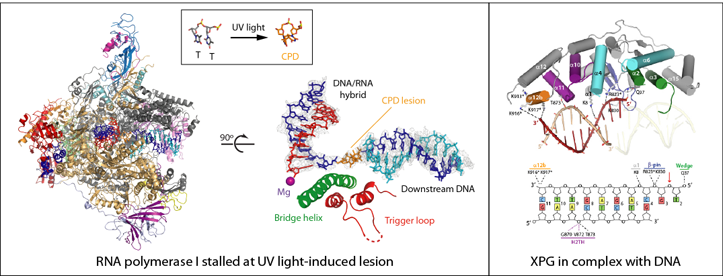
DNA damage threatens cell life and must be repaired to maintain genome integrity. Transcription is particularly sensitive to DNA damage and, hence, it is intimately linked to DNA repair. Different lesions induce RNA polymerase stalling, which constitutes a signal for recruitment of DNA repair factors. Along this process, TFIIH maintains a repair bubble that is incised by two endonucleases, XPF and XPG, to eliminate the lesion. Alterations in these repair factors associate with genetic disorders such as xeroderma pigmentosum and Cockayne syndrome, with symptoms ranging from skin cancer risk (“children of the moon”) to mental retardation.
We used cryo-EM and functional activity tests to study the mechanism for the recognition of UV light-induced DNA lesions by Pol I, which provides the first clues on how ribosomal DNA is protected from harmful lesions [PNAS, 2018]. In addition, we used X-ray crystallography to determine the structure of the XPG endonuclease catalytic core, which uncovered unique features of this enzyme that allow incision of damaged DNA repair bubbles [NAR, 2020].