The Myeloid Cell Biology group led by Dr. Ángel Corbí at Centro de Investigaciones Biológicas, in which Dr. Ángeles Domínguez Soto is integrated, actively studies the molecular mechanisms involved in the acquisition of pro- and anti-inflammatory functions of macrophages in order to identify therapeutic targets for inflammatory and autoimmune diseases. Recently, Dr. Ángeles Domínguez Soto, in collaboration with the group led by Dr. Amaya Puig Kröger at the Hospital Gregorio Marañón, has contributed to the identification of a new mechanism of action of methotrexate in macrophages.
Rheumatoid arthritis (RA) is a chronic and systemic inflammatory disease that affects the synovial joints. In patients with RA, the macrophages in the joints present a pro-inflammatory profile and are the main producers of the pro-inflammatory cytokines responsible for the pathogenesis of RA. In fact, the number of macrophages in the synovial fluid constitutes a biomarker of the response to treatment in patients with RA.
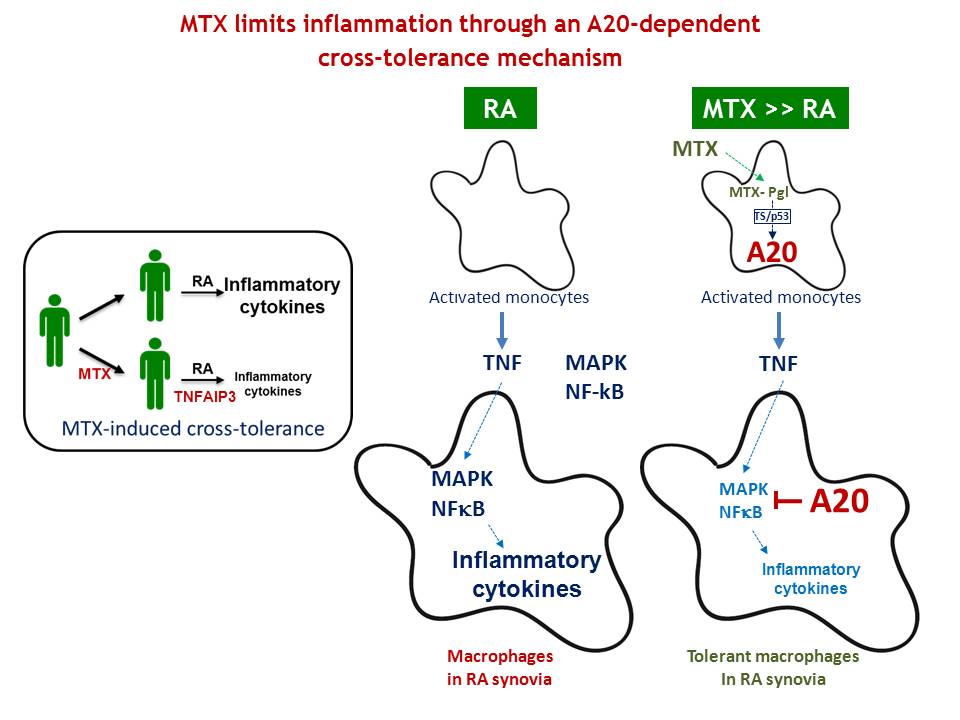
Methotrexate (MTX) is the reference drug for the treatment of RA. Despite its therapeutic use for more than 25 years, the mechanisms through which it exerts its anti-inflammatory action are not completely known. In the work published in the Journal Annals of the Rheumatic Diseases, Domínguez-Soto and colleagues describe the molecular mechanism of the anti-inflammatory activity of methotrexate administered at low doses. Specifically, the study has shown that MTX conditions the macrophages towards a tolerant state, decreasing the production of pro-inflammatory cytokines induced by pathogens, tumor necrosis factor (TNF) and even the synovial fluid of patients with RA.
Using a combination of transcriptomic, biochemical and functional approaches, the study revealed that MTX increases the expression of the TNFAIP3 gene, which encodes for the A20 protein, a negative regulator of NFκB signaling that has been described as a gene of susceptibility to RA. In fact, and using siRNA silencing strategies, it has been possible to determine that the elimination of A20 considerably reduces the tolerance-inducing effect of MTX. The pathophysiological significance of these results is supported by the analysis carried out on samples from RA patients, in which it is proven that TNFAIP3 expression in peripheral blood cells is significantly higher in those RA patients who respond to treatment with MTX.
The determination of the ability of MTX to limit the pro-inflammatory response of macrophages through the increase of the A20 protein levels, carried out by Dr. Ángeles Domínguez et al., has a great relevance since it positions this protein as a biomarker of response to Methotrexate with clinical utility.
Reference: Methotrexate limits inflammation through an A20-dependent cross-tolerance mechanism. Dominguez-Soto Á, Municio C, Fuentelsaz-Romero S, Lamana A, Montes N, Cuevas VD, Campos RG, Pablos JL, González-Álvaro I, Puig-Kröger A. Ann Rheum Dis. 2018 Feb. doi: 10.1136/annrheumdis-2017-212537.

